
Therefore, it is confirmed that electrons do not exist inside the nucleus. Second reason that electron can not exist inside the nucleus is that experimental results show that no electron or particle in the atom possess energy greater than 4 MeV. Heisenberg: The Uncertainty Principle (Modern Plays) Stephens, Simon on. However, it is observed that beta-particles (electrons) ejected from the nucleus during b –decay have energies of approximately 3 Me V, which is quite different from the calculated value of 19.6 MeV. 
Therefore, if the electron exists in the nucleus, it should have an energy of the order of 19.6 MeV. This relation expresses Heisenberg’s uncertainty principle. The product of the uncertainty in position of a particle and the uncertainty in its momentum can never be less than one-half of the reduced Planck constant: (7.3.1) x p 2. If this is p the uncertainty in the momentum of electron ,then the momentum of electron should be at least of this order, that is p=1.05*10 -20 kg m/sec.Īn electron having this much high momentum must have a velocity comparable to the velocity of light. Definition: The Heisenberg’s Uncertainty Principle. If electron is to exist inside the nucleus, then uncertainty in the position of the electron is given by 
As the radius of the nucleus in approximately 10 -14 m. First, we note that these patterns are identical, following d sin θ = mλ d sin θ = mλ size 12.In this article, we will prove that electrons cannot exist inside the nucleus.īut to prove it, let us assume that electrons exist in the nucleus. Trying to pin a thing down to one definite position will make its momentum less well pinned down, and vice-versa. Werner Heisenberg stumbled on a secret of the universe: Nothing has a definite position, a definite trajectory, or a definite momentum. uncertainty principle, physical principle, enunciated by Werner Heisenberg in 1927, that places an absolute, theoretical limit on the combined accuracy of. 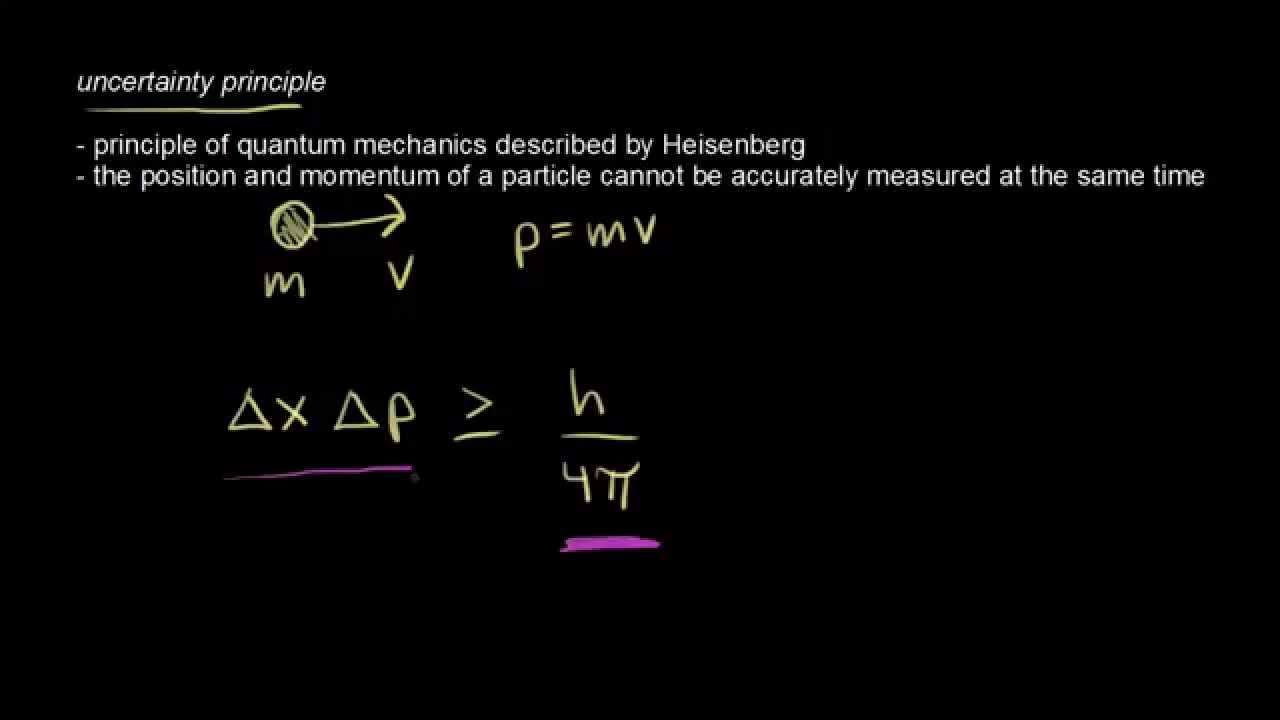
Consider the double-slit patterns obtained for electrons and photons in Figure 29.25. The Uncertainty principle is also called the Heisenberg uncertainty principle. Let us explore what happens if we try to follow a particle. It is somewhat disquieting to think that you cannot predict exactly where an individual particle will go, or even follow it to its destination. Those who developed quantum mechanics devised equations that predicted the probability distribution in various circumstances. There is a certain probability of finding the particle at a given location, and the overall pattern is called a probability distribution. After compiling enough data, you get a distribution related to the particle’s wavelength and diffraction pattern. However, each particle goes to a definite place (as illustrated in Figure 29.24). The idea quickly emerged that, because of its wave character, a particle’s trajectory and destination cannot be precisely predicted for each particle individually. Review the importance and background of the principle, and. Both patterns are probability distributions in the sense that they are built up by individual particles traversing the apparatus, the paths of which are not individually predictable.Īfter de Broglie proposed the wave nature of matter, many physicists, including Schrödinger and Heisenberg, explored the consequences. The Heisenberg uncertainty principle is a law in quantum mechanics that addresses limitations in the accuracy of measuring two variables. Figure 29.25 Double-slit interference for electrons (a) and photons (b) is identical for equal wavelengths and equal slit separations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
